Selection of Stainless Steel Suitable for Environment
Stainless steel is short for stainless acid-resistant steel. Weak corrosive media such as air, steam, water or the steel with rust resistance is called stainless steel.
Each stainless steel has good properties in its specific application fields. The key to success is first to clarify the usage, and then to determine the correct steel grade.
Here we will analyze how to choose stainless steel suitable for usage environment.
1. What is stainless steel?
2. How many kinds of stainless steel?
3. How to choose suitable stainless steel?
1. What is stainless steel?
Stainless steel is an alloy of Iron with a minimum of 10.5% Chromium. Chromium produces a thin oxide layer on the surface of steel, which is called passive layer. This prevents any further corrosion of the surface. Increasing the amount of chromium gives an increased resistance to corrosion.
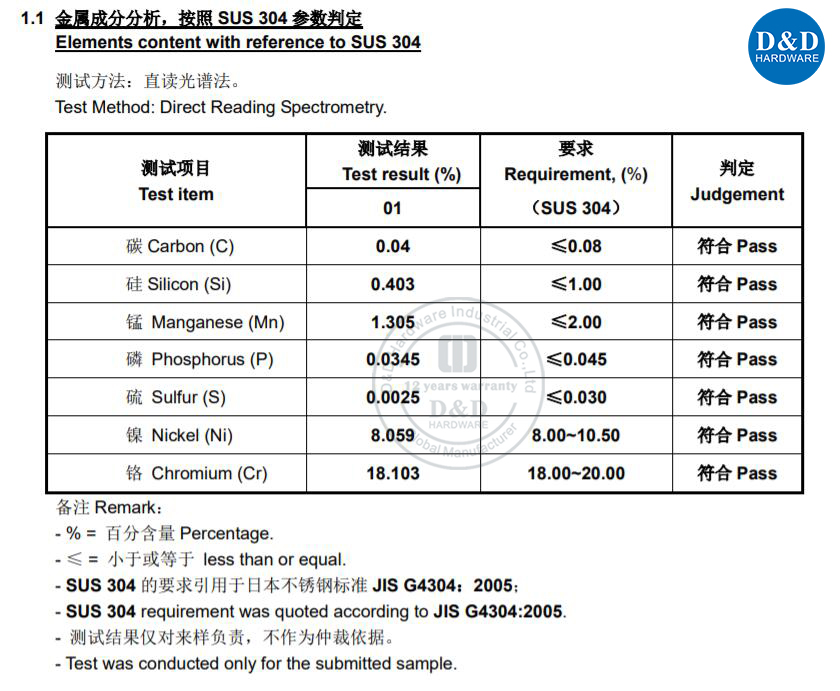
![Elements content with reference to SUS 304. Elements content with reference to SUS 304.]()
Stainless steel also contains different amounts of carbon, silicon and manganese. Other elements such as nickel and molybdenum may be added to impart other useful properties such as enhanced formability and increased corrosion resistance.
The corrosion resistance of stainless steel decreases with the increase of carbon content. Therefore, the carbon content of most stainless steels is low, a maximum of 1.2% carbon by mas, and the (carbon content) of some steels is even lower than 0.03% (such as 00Cr12). The main alloying element in stainless steel is chromium. Only when the content of chromium reaches a certain value, the steel has corrosion resistance. Therefore, the general content of Cr (chromium) in stainless steel is at least 10.5%. The stainless steel also contains Ni, Ti, Mn, N, Nb, Mo, Si, Cu and other elements.
Due to the different usage of products, their processing technology and quality requirements of raw materials are also different.
2. How many kinds of stainless steel?
Stainless steel is usually divided into martensitic steel, ferritic steel, austenitic steel, austenitic-ferritic (duplex) stainless steel and precipitation hardening stainless steel.
According to the composition, it can be divided into chromium stainless steel, chromium-nickel stainless steel and chromium-manganese-nitrogen stainless steel. There is also special stainless steel for pressure vessels.
3. How to choose suitable stainless steel?
By understanding the corrosion mode, it can also be used as a reference for choosing stainless steel types.
General Corrosion - Stainless steel will not corrode as uniformly as ordinary carbon steel and alloy steel. However, for certain chemicals, especially acids, depending on concentration and temperature, the passivation layer may be uniformly eroded. Certain high concentrations of hydrochloric acid and sulphuric acid are particularly corrosive to stainless steel.
Pitting corrosion - The passivation layer on stainless steel will be corroded by some chemicals. In daily materials such as salts and bleaching agents, pitting can be avoided by ensuring that stainless steel does not have long-term exposure to harmful chemicals or by selecting a more corrosion-resistant steel grade.
Crevice corrosion - stainless steel is supplied with oxygen, and passivation layer can be formed on the surface. In the narrow gap, oxygen cannot always touch the surface of stainless steel, making the stainless steel susceptible to erosion. A more corrosion resistant grade is required to avoid crevice corrosion.
Stress corrosion cracking (SCC) - This is a relatively rare form of corrosion that requires a special combination of tensile stress, temperature and corrosive substances (usually chloride ions). Frequently occurring environments are hot water tanks and swimming pools. Another form, called sulfide stress corrosion cracking (SSCC), is related to hydrogen sulfide in oil and gas exploration and production.
Intergranular corrosion - This is a very rare form of corrosion. If the carbon content in steel is too high, chromium can combine with carbon to form chromium carbide. This happens when the welding process between 450 and 850 degrees Celsius. This can be avoided by choosing low carbon grades (so-called "L" grades) or using titanium or niobium steels (preferred carbon bonding).
Galvanic corrosion - If two different metals are in contact with each other or contact with electrolytes (such as water or other solutions), corrosion occurs. It can be avoided by separating metals with non-metallic insulators such as rubber.
Stainless steel is widely used for its durability and virtually maintenance-free surface properties. Stainless steel can be used in furniture, transportation, chemistry, medical treatment, construction, etc. D&D Hardware sells high-quality stainless steel hinge, mortise lock, door handle, door bolt and other door hardware products, widely used in terminal, hotel, hospital, educational institutions, etc. We also provide complete product supporting solutions to solve customer queries.
For further information about stainless steel hardware or any of our services, click here.
Contact us Now:
Contact: David Jian
Mob:0086-139 2903 7292
Email: David@dndhardware.com, sales@dndhardware.com,
Jobby Zhang
Mob:0086-137 2599 9617
Email: jobby@dndhardware.com


 English
English